|
1/9/2023 0 Comments Sp3d hybrid orbitals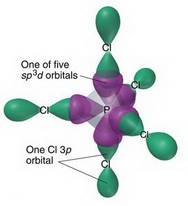 
Some transargononic structures of metal sulfides have been proposed recently by Franzen (1966). Ī transargononic structure for sulfur, with six bonds formed by sp3d2 hybrid orbitals, was suggested for sulfur in the octahedral molecule SF6 long ago, and also for one of the sulfur atoms, with ligancy 6, in binnite (Pauling and Neuman, 1934). These identical orbitals point toward the six corners of a regular octahedron. We need six orbitals to accommodate six electron pairs around an atom in an octahedral arrangement, as in SF6 and XeF4, and so we need to use two d-orbitals in addition to the valence s- and p-orbitals to form six sp3d2 hybrid orbitals (Fig.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
